
Thus all atoms are not the same which can be defined by the different properties of the substances. They differ in mass, size and other chemical properties. If we compare two elements carbon and hydrogen. A neutral atom will have the same number of electrons as protons. Even the atoms of the same element are different due to the difference in the number of neutrons like in isotopes. This means that all atoms of the same element. The carbon atoms in a diamond have a tetrahedral structure around each. All atoms of the same element have the same number of protons, which is its atomic number. A piece of an element that we are able to see or handle is made of many, many atoms and all atoms are the same. For example, a diamond, coal and soot all are allotropes of carbon. Isotopes are atoms of the same element that have different numbers of neutrons but the same number of protons and electrons. Allotropes may have different physical appearances and different characteristics such as electrical conductivity. The other elements have been produced artificially or as a result of nuclear reactions.Ītoms of the same element that are arranged in different molecular formations are called allotropes. Of these elements, only the first 98 are naturally occurring. There are 118 known elements, arranged in the periodic table in order of atomic number. 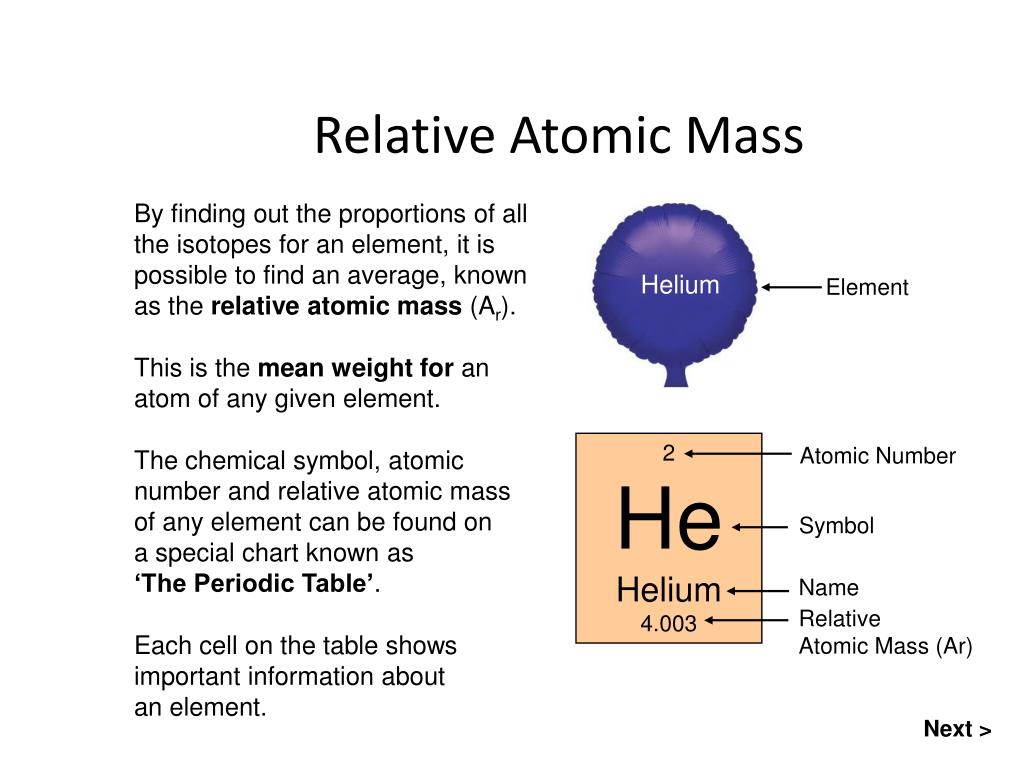
Isotopes have the same atomic number but different atomic masses.Ītoms of an element share that element’s chemical and physical properties, such as boiling point, melting point and stability.Īn element is the simplest form of matter and cannot be broken down further by chemical means. Atoms of the same element that have a different number of neutrons are called isotopes. Atoms of an element usually have the same number of neutrons as protons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
